 In order for the question and also answer for the question. So this is the region they are for your our order richard barium which is greater than calcium, which is better than silicon then which is greater than oxygen. 
What nucleus here it is nuclear judge which is increases the rate at which is what increases. Harry reid Because he called you the nucleus here nucleus. So here the atomic radius which decreases in the periodic table to move what's left to ride its region. And I think that your question initially meant to compare the size of Al3+ NOT Al3 (largely unknown). potassium should have the biggest atomic radius, followed by selenium, then bromine.i.e. Talk to down because there are moved cell of the regulatory originally because what it's regeneration these are record teach are what move from here to promote the sales. For selenium, potassium, and bromine, we gots n 4. Right periodic increasing the period irritable move to move towards top two down record. So here the atomic radius which increases the record the atomic what we regard the atomic radius, your record actor the ideas which is what which is increases so you're like that which is increase in the hot periodic in the harry writer periodic here. The boundary of the I looked on her right boundary boundary off the dock the record boundary of the word electron. The general trend is that atomic sizes increase as one moves downwards in the Periodic Table of the Elements, as electrons fill outer electron shells. From the above arrangement of elements with their atomic radii, we find that the atomic radius (or atomic size) decreases on moving from left to right in. Jihad is a dish transform the center of the nucleus is greater distance from the record distance from the world from the centers of the regular center of what center of the nucleus center of the nucleus. Atomic the ideas the atomic radius is this site of religious site of what atoms then you're the atomic ideas should reiterate the atomic the radios. So we know atomic radios jihad size of autumn. So it is the largest side then it thus more left side. Which is then what silicon then which is better than oxygen. Arrange the elements according to atomic radius. Start your trial now First week only 4.99 arrow.  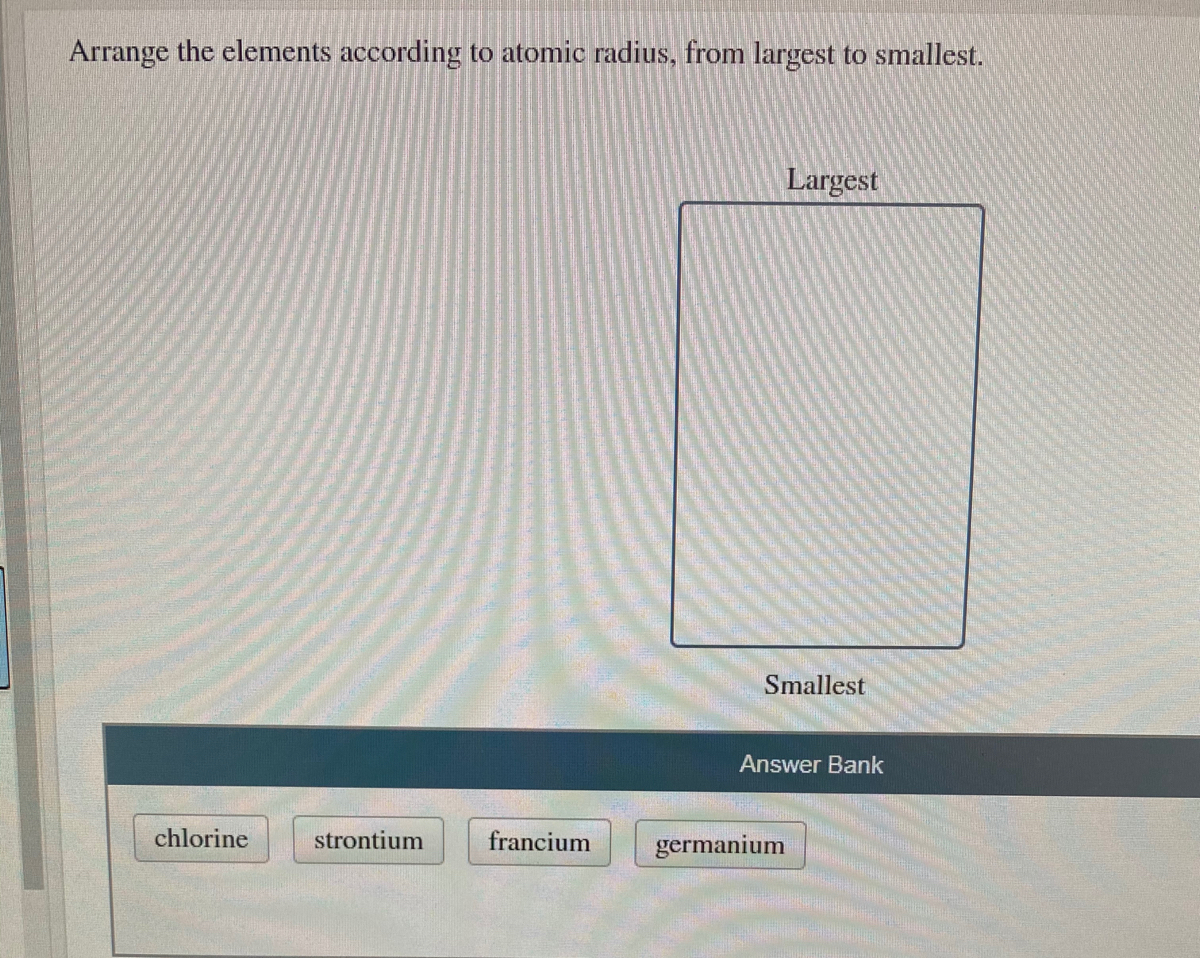
Largest radius Na Ar Mg Al Si P S CI Smallest radius Answer Bank Incorrect. So you are the gibbon compound which are here right here shoulders which is a barium record barium then which is which is greater than calcium which is better than calcium which is better than words silicon. Solution for Arrange the elements according to atomic radius. From what largest to record the largest too smallest. Question: Arrange the elements according to atomic radius, from largest to smallest. We have been questioned that he jacks here we arranged the elements according to their atomic radius. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
